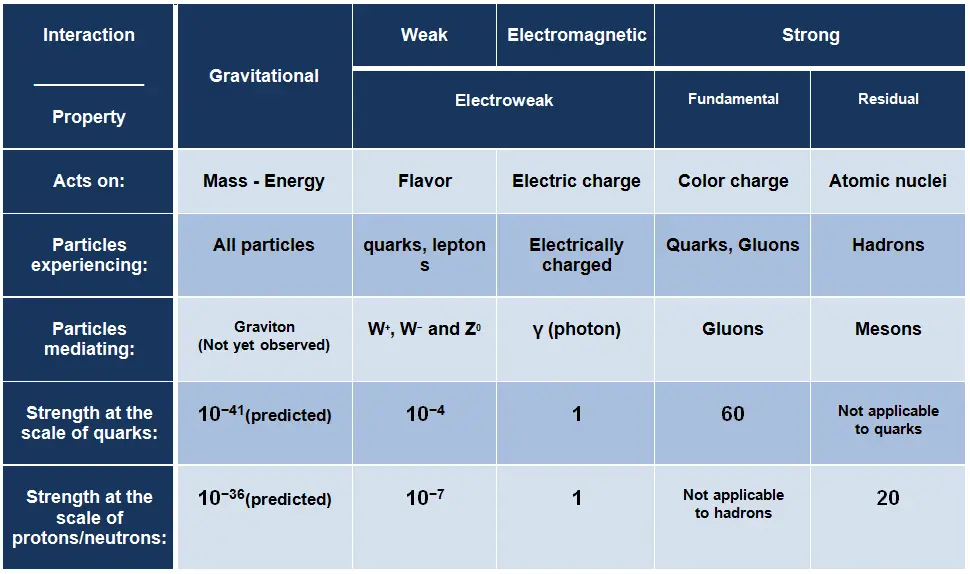
Electromagnetic Interaction – Electromagnetic Force
The electromagnetic force is the force responsible for all electromagnetic processes. It acts between electrically charged particles. It is infinite-ranged force, much stronger than gravitational force, obeys the inverse square law, but neither electricity nor magnetism adds up in the way that gravitational force does. Since there are positive and negative charges (poles), these charges tend to cancel each other out. Electromagnetism includes the electrostatic force acting between charged particles at rest, and the combined effect of electric and magnetic forces acting between charged particles moving relative to each other.
The photon, the quantum of electromagnetic radiation, is an elementary particle, which is the force carrier of the electromagnetic force. Photons are gauge bosons having no electric charge or rest mass and one unit of spin. Common to all photons is the speed of light, the universal constant of physics. In empty space, the photon moves at c (the speed of light – 299 792 458 metres per second).
Forces between static electrically charged particles are governed by the Coulomb’s law. Coulomb’s Law can be used to calculate the force between charged particles (e.g. two protons). The electrostatic force is directly proportional to the electrical charges of the two particles and inversely proportional to the square of the distance between the particles. Coulomb’s Law is stated as the following equation.
Both, the Coulomb’s law and the magnetic force, are summarized in the Lorentz force law. Fundamentally, both magnetic and electric forces are manifestations of an exchange force involving the exchange of photons.
The electromagnetic force plays a major role in determining the internal properties of most objects encountered in daily life. The chemical properties of atoms and molecules are determined by the number of protons, in fact, by number and arrangement of electrons.
Gravitational Interaction – Gravitational Force
Gravity was the first force to be investigated scientifically. The gravitational force was described systematically by Isaac Newton in the 17th century. Newton stated that the gravitational force acts between all objects having mass (including objects ranging from atoms and photons, to planets and stars) and is directly proportional to the masses of the bodies and inversely proportional to the square of the distance between the bodies. Since energy and mass are equivalent, all forms of energy (including light) cause gravitation and are under the influence of it. The range of this force is ∞ and it is weaker than the other forces. This relationship is shown in the equation below.
The equation illustrates that the larger the masses of the objects or the smaller the distance between the objects, the greater the gravitational force. So even though the masses of nucleons are very small, the fact that the distance between nucleons is extremely short may make the gravitational force significant. The gravitational force between two protons that are separated by a distance of 10-20 meters is about 10-24 newtons. Gravity is the weakest of the four fundamental forces of physics, approximately 1038 times weaker than the strong force. On the other hand, gravity is additive. Every speck of matter that you put into a lump contributes to the overall overall gravity of the lump. Since it is also a very long range force, it is dominant force at the macroscopic scale, and is the cause of the formation, shape and trajectory (orbit) of astronomical bodies.
We hope, this article, Electromagnetic Force vs Gravitational Force, helps you. If so, give us a like in the sidebar. Main purpose of this website is to help the public to learn some interesting and important information about radiation and dosimeters.