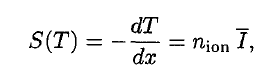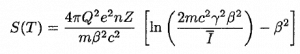In radiation protection, the committed dose is a dose quantity that measures the stochastic health risk due to an intake of radioactive material into the human body. Commited dose is given the symbol E(t), where t is the integration time in years following the intake. The SI unit of E(t) is the sievert (Sv) or but rem (roentgen equivalent man) is still commonly used (1 Sv = 100 rem). Unit of sievert was named after the Swedish scientist Rolf Sievert, who did a lot of the early work on dosimetry in radiation therapy.
Committed dose allows to determine the biological consequences of irradiation caused by radioactive material, that is inside our body. A committed dose of 1 Sv from an internal source represents the same effective risk as the same amount of effective dose of 1 Sv applied uniformly to the whole body from an external source.
As an example, let assume an intake of radioactive tritium. For tritium, the annual limit intake (ALI) is 1 x 109 Bq. If you take in 1 x 109 Bq of tritium, you will receive a whole-body dose of 20 mSv. Note that, the biological half-life about 10 days, while the radioactive half-life is about 12 years. Instead of years, it takes a couple of months until the tritium has been pretty well eliminated. The committed effective dose, E(t), is therefore 20 mSv. It does not depend whether a person intakes this amount of activity in a short time or in a long time. In every case, this person gets the same whole-body dose of 20 mSv.
The ICRP defines two dose quantities for individual committed dose.
Committed Effective Dose
According to the ICRP, the committed effective dose, E(t) is defined as:
“The sum of the products of the committed organ or tissue equivalent doses and the appropriate tissue weighting factors (wT), where t is the integration time in years following the intake. The commitment period is taken to be 50 years for adults, and to age 70 years for children.”
Committed Equivalent Dose
According to the ICRP, the committed equivalent dose, HT(t) is defined as:
“The time integral of the equivalent dose rate in a particular tissue or organ that will be received by an individual following intake of radioactive material into the body by a Reference Person, where t is the integration time in years.”
Special Reference: ICRP, 2007. The 2007 Recommendations of the International Commission on Radiological Protection. ICRP Publication 103. Ann. ICRP 37 (2-4).
Internal Dose Uptake
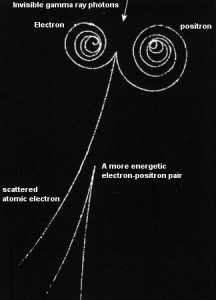
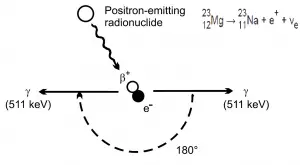
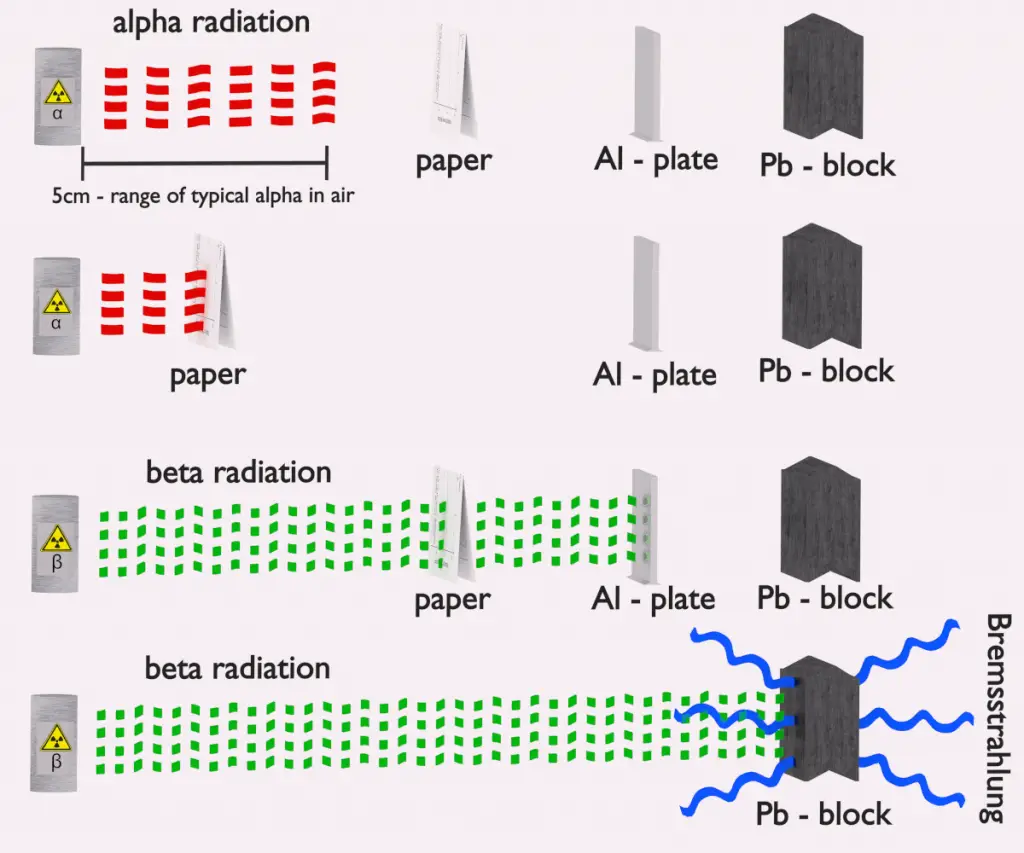
If the source of radiation is inside our body, we say, it is internal exposure. The intake of radioactive material can occur through various pathways such as ingestion of radioactive contamination in food or liquids, inhalation of radioactive gases, or through intact or wounded skin. Most radionuclides will give you much more radiation dose if they can somehow enter your body, than they would if they remained outside.
But when a radioactive compound enters the body, the activity will decrease with time, due both to radioactive decay and to biological clearance. The decrease varies from one radioactive compound to another. For this purpose, the biological half-life is defined in radiation protection.
The biological half-life is the time taken for the amount of a particular element in the body to decrease to half of its initial value due to elimination by biological processes alone, when the rate of removal is roughly exponential. The biological half-life depends on the rate at which the body normally uses a particular compound of an element. Radioactive isotopes that were ingested or taken in through other pathways will gradually be removed from the body via bowels, kidneys, respiration and perspiration. This means that a radioactive substance can be expelled before it has had the chance to decay.
As a result, the biological half-life significantly influences the effective half-life and the overall dose from internal contamination. If a radioactive compound with radioactive half-life (t1/2) is cleared from the body with a biological half-life tb, the effective half-life (te) is given by the expression:
As can be seen, the biological mechanisms always decreases the overall dose from internal contamination. Moreover, if t1/2 is large in comparison to tb, the effective half-life is approximately the same as tb.
For example, tritium has the biological half-life about 10 days, while the radioactive half-life is about 12 years. On the other hand, radionuclides with very short radioactive half-lives have also very short effective half-lives. These radionuclides will deliver, for all practical purposes, the total radiation dose within the first few days or weeks after intake.
For tritium, the annual limit intake (ALI) is 1 x 109 Bq. If you take in 1 x 109 Bq of tritium, you will receive a whole-body dose of 20 mSv. The committed effective dose, E(t), is therefore 20 mSv. It does not depend whether a person intakes this amount of activity in a short time or in a long time. In every case, this person gets the same whole-body dose of 20 mSv.
We hope, this article, Committed Dose – Committed Effective Dose, helps you. If so, give us a like in the sidebar. Main purpose of this website is to help the public to learn some interesting and important information about radiation and dosimeters.